|
8/27/2023 0 Comments Sigma bond example 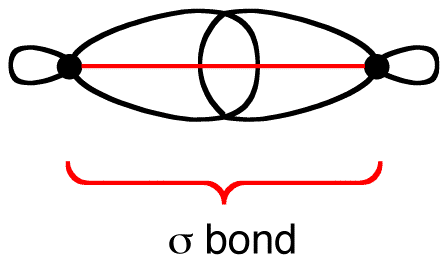
This is why in organic chemistry, you will see reactions involving breaking pi bonds far more often than sigma bonds as they are broken much easier.Īn interesting feature distinguishing sigma and pi bonds is the former is characterized by free rotation about the bond axis, while the presence of a pi bond restricts this, and the atoms are locked in their position relative to the double bond. The side-to-side orbital overlap allows a less efficient electron sharing, and therefore, p bonds are generally weaker than sigma bonds. * The angle between the plane and p orbitals is 90 o. * All the atoms on the double bond are in one plane. Here are some key parameters about the sp 2 hybridization and double bonds that you need to know: In each double bond, there is one sigma and one π bond. So, the two carbons in ethylene, which is the first member of the alkene family, are double-bonded. The two carbons are sp 2-hybridized where the sp 2 orbitals overlap to form the sigma bond, and the unhybridized p orbitals form the pi bond. The pi bond is formed by a side-to-side overlap of two p orbitals provided by adjacent atoms. On the other hand, in ethene, or ethylene (C 2H 4), there is one sigma and one pi bond between the two carbon atoms: For example, the single bond between the two carbons in ethane (C 2H 6) is a sigma bond because it is formed by overlapping two sp 3 orbitals of the adjacent carbon atoms: In short, you can remember that single bonds are sigma bonds. The bonds that form by the head-on overlap of orbitals are called σ (sigma) bonds because the electron density is concentrated on the axis connecting the two atoms. Now, there are two types of covalent bonds: sigma ( σ) and pi (π) bonds. The bonds that are not shown are assumed to be bonds to hydrogen.We mentioned in the previous post that covalent bonds are formed as a result of sharing two valence electrons in overlapping orbitals of two atoms.įor example, the following Lewis structures represent covalent bonds together with some lone pairs of electrons: Line angle representation for propane equivalent to or CH3CH2CH3 The arrows point to the positions of the carbon atoms.Įvery carbon atom has to form 4 bonds. The beginning and the end of the zig-zag line, as well as any breaks in direction represent carbon atoms. The resulting representations are known as line-angle formulas. 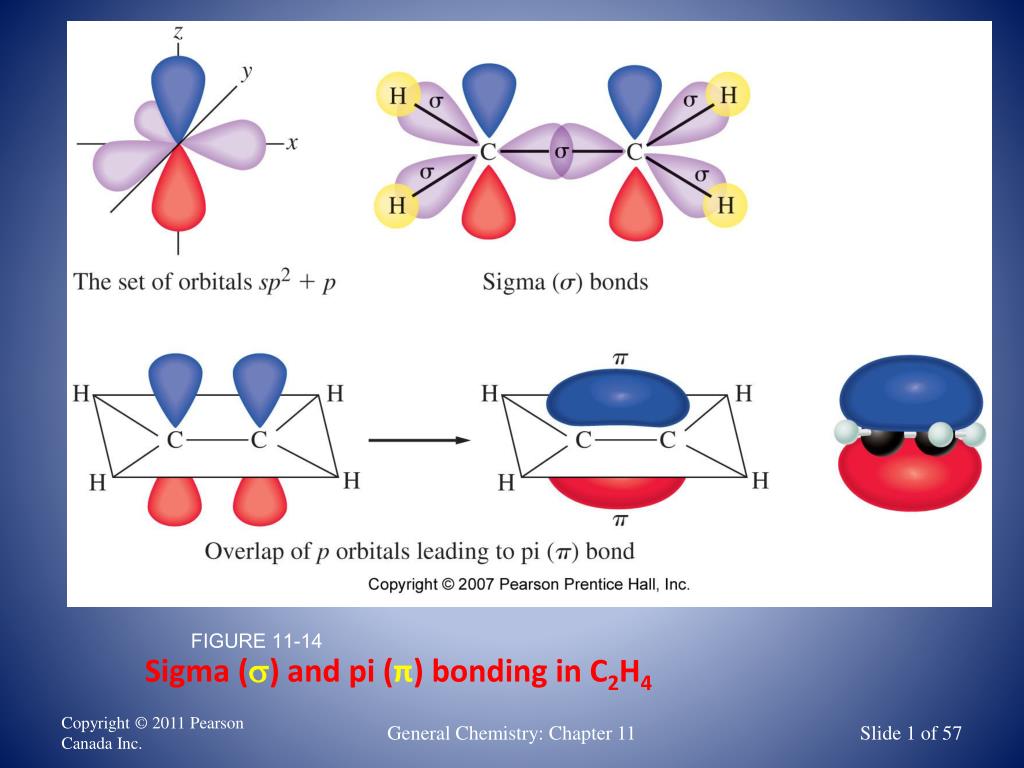
A short hand notation that uses zig-zag lines has been developed. Writing Lewis formulas, or even condensed formulas, for alkanes of many carbon atoms can quickly become cumbersome. Two representations of propane, where the zig-zag structure of the carbon chain becomes apparent In alkanes of 3 carbon atoms or more, the main carbon chain acquires a zig-zag structure due to the 109.5 o angle between C–C bonds, such as in propane: It can be easily seen that the only type of covalent bonds present in alkanes are sigma bonds, also loosely known as single bonds. Two sp 3 carbons can also overlap to form a C–C sigma bond where two sp 3 orbitals overlap head to head, such as in the formation of the ethane molecule: The bonds between the sp 3 orbitals of hybridized carbon and the s orbitals of hydrogen in methane are also example of sigma bonds. The bond between two hydrogen atoms is an example of sigma bonding. When orbitals approach each other in a head to head fashion, the resulting covalent bonds are called sigma bonds.Īs illustrations, consider the bonds that have already been studied. We’ll discuss sideways overlap later in connection with alkenes and alkynes, that is, hydrocarbons that have double and triple bonds respectively. For now, however, we are concerned only with head to head overlap because that’s the only type that occurs in alkanes. p-orbitals, on the other hand, can approach each other either sideways or head to head. Hybrid orbitals also undergo mostly head to head overlap when forming covalent bonds. Only head to head overlap is possible with s orbitals because they are spherical. When atomic orbitals (pure or hybrid) of different atoms overlap to form covalent bonds, they may approach each other in two major ways: head to head, or sideways. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
